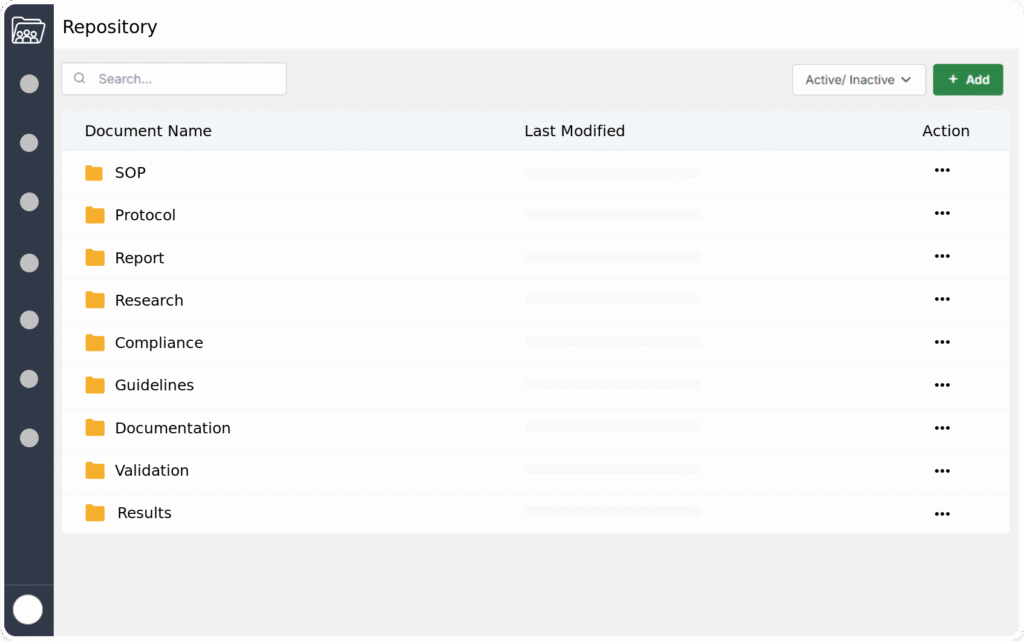











Utilizes standardized templates to ensure consistency in regulatory documents, such as SOPs and other documents. This minimizes formatting errors and adheres to industry guidelines.
Manages documents from creation to retirement, ensuring compliance at every stage. This includes efficient handling of drafting, review, approval, and archiving processes. Streamlined lifecycles help maintain accountability and regulatory adherence.
A comprehensive app that can be used immediately.
Uses a tagging system to categorize documents for easier retrieval based on type, project, or requirements. This simplifies the organization and access of critical information. Efficient tagging supports compliance by enhancing document management practices.
Controls access to folders and files based on user permissions to protect sensitive information. This ensures that only authorized personnel can view or edit critical documents. Strict access management enhances data security and supports regulatory compliance.
Provides an easy way to search for documents and content, allowing users to quickly find what they need. This feature saves time and makes it simple to access important information.
First light to twilight, we’re your weekday lifeline, spanning emails, calls, chats, and remote magic.
Progressively innovate models and offer unique benefits for diverse integrated markets.
Receive immediate notifications that ensure you’re intricately linked to the rhythm of ongoing events.